DNMT3A gene mutations are more frequent in cytogenetically normal acute myeloid leukemia (AML) and frequently associated with NPM1 mutation and internal tandem duplication (ITD) of the FLT3 gene. However, DNMT3A mutation is not included as a distinct prognostic group in the recent European Leukemia Net (ELN) 2022 genetic risk classification of AML. In the context of allogeneic hematopoietic cell transplantation (allo-HCT), the prognostic value of DNMT3A is still not well studied. We evaluated, through the European Society for Blood and Marrow Transplantation (EBMT) registry, the impact of DNMT3A mutation, according to the karyotype, and NPM1 and FLT3-ITD mutation status, on post-transplant outcomes in AML patients receiving allo-HCT in first complete remission (CR1).
We identified a total of 1374 adult AML patients (51% female, median age 57 years; range 18-78 years) with ELN-2022 intermediate- or poor-risk cytogenetics and available DNMT3A, FLT3ITD and NPM1 mutation status, who underwent their first allo-HCT in CR1 between 2013 and 2021. Of these, 631 (46%) patients had DNMT3A mutation. Patients with mutant DNMT3A were more likely to be female (57% versus 46%, p<0.001), to have normal cytogenetics (75% versus 54%, p<0.001), FLT3-ITD co-mutation (50% vs 23%, p<0.001), NPM1 co-mutation (60% vs 21%, p<0.001), and to receive a myeloablative conditioning and in vivo T cell depletion. After a median follow-up of 2.3 years, the 2-year leukemia free survival (LFS) and overall survival (OS) were 62% and 69%, respectively. The subsequent analysis was split according to baseline karyotype (normal or abnormal).
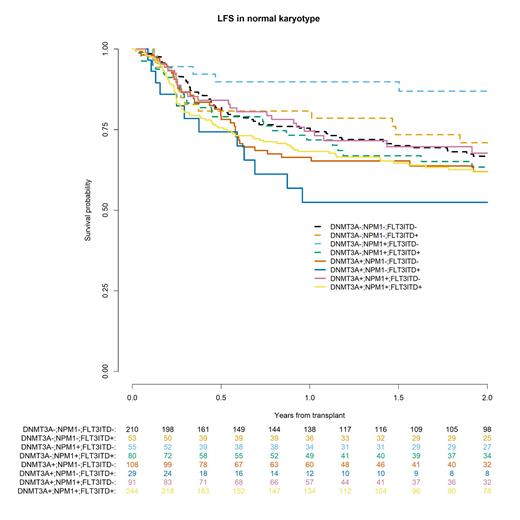
Among 870 patients with normal cytogenetics, 472 (54%) patients had DNMT3A mutation. The 2-year relapse incidence (RI), LFS and OS were 24%, 63% and 70% respectively, in patients with DNMT3A mutation compared to 18%, 69% and 74% in patients with wild type DNMT3A. Among these patients with normal karyotype, multivariable analyses (MVA) were done within the 4 groups of FLT3-ITD and NPM1 mutations. Within the group of 324 patients with both FLT3-ITD and NPM1 positives, 244 were triple-positive (75%), LFS and OS were not significantly different with and without DNMT3A mutation (2y LFS: 62% vs 63%, HR: 0.98, p=0.95; 2y OS: 70% vs 73%, HR: 1.17, p=0.58). Conversely, among 146 patients with NPM1 mutation without FLT3-ITD, LFS was significantly lower for 91 patients with DNMT3A mutation (2y LFS: 68% vs 87%, HR: 2.70, p=0.02) but OS was not significantly different (2y OS: 79% vs 87%, HR: 2, p=0.13). On the other hand, among 318 with wild type NPM1 and without FLT3-ITD, LFS and OS were not significantly different for 108 patients with DNMT3A mutation compared to 210 patients with wild type DNMT3A (2y LFS: 62% vs 67%, HR: 1, p=0.98; 2y OS: 65% vs 71%, HR: 1.02, p=0.94). Conversely, among 82 patients with FLT3-ITD and wild type NPM1, 2-year LFS and OS were 53% and 58% respectively, for 29 patients with DNMT3A mutation compared to 71% and 76% respectively, for 53 patients with wild type DNMT3A. The number of patients didn't allow to perform MVA in this group.
Finally, among 504 patients with abnormal cytogenetics, 159 (31%) patients had DNMT3A mutation. On MVA, DNMT3A mutation did not significantly affect LFS or OS. Poor-risk cytogenetics was associated with worse LFS, OS and increased RI. Older age negatively affected LFS and OS.
In conclusion, in AML patients allografted in CR1, the impact of DNMT3A mutation depends on the karyotype and co-occurring mutations. Our data suggest that DNMT3A mutation negatively affects post-transplant survival selectively in patients with normal karyotype and either NPM1 mutation without FLT3-ITD, or patients with normal karyotype, FLT3-ITD and wild type NPM1. DNMT3A mutation shows no impact on post-transplant outcomes in other settings.
Disclosures
Huynh:Medac: Other: Advisory board; Astellas: Other: Advisory board; Pfizer: Other: advisory board; Jazz: Other: travel fees, advisory board; Servier: Other: Advisory board; Neovii: Other: Advisory board; Novartis: Other: travel fees, advisory board. Mayer:MSD: Research Funding; Novartis: Research Funding. Esteve:Jazz Pharmaceuticals: Consultancy, Research Funding; Abbvie: Consultancy; Gilead: Consultancy; Kronos Bio: Research Funding; Pfizer: Research Funding; Astellas: Consultancy. Ciceri:ExCellThera: Other: Scientific Advisory Board . Mohty:JAZZ PHARMACEUTICALS: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal